Sulfur has six valence electrons, however each fluorine actually has 7 valence electrons a piece it is a group 7 atom. The question says, "SF2 1-" which I take to mean one sulfur, two fluorines and a charge of Pauli, as in Wolfgang Pauli, was a pioneer in quantum theory. Hemiketal is having trouble counting electrons, too.
#DRAW THE LEWIS STRUCTURE FOR SF2 HOW TO#
Dream is a little unclear about how to draw Lewis structures. It has a bent geometry with single bonds to each fluorine and two lone pairs on the sulfur.

There does exist a neutral sulfur difluoride molecule. Assuming the anion exists, it would have 21 electrons, six for sulfur and seven each for the fluorine atoms, plus one more because of its "charge". You seem to have trouble counting electrons. Fluorine has 7 valence electrons, not six. Asked in Chemical Bonding How do you draw a Lewis dot structure for an ionic compound? How do you draw a lewis dot structure for an ionic compound?For one thing, it would be a free radical, meaning that it has an odd number of electrons.įree radicals tend to be quite reactive because of the single unpaired electron. Lewis structure, electron dot diagram, electron dot structure It looks like a lewis dot structure for C2H2F2. The Lewis dot structure would be Cr with one dot over it. SF2 can be represented by the Lewis dot structure of Call for the answer! Asked in Elements and Compounds What is Lewis dot structure of sodium sulphate?
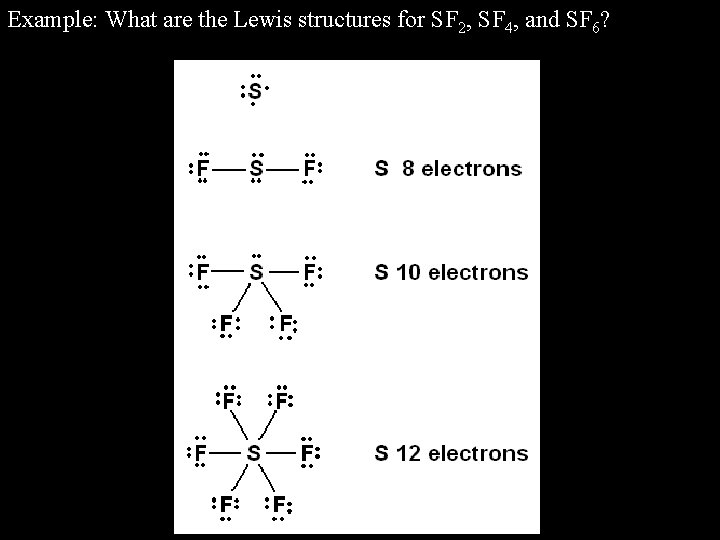
Sulphur difluoride is the chemical name for the molecular formula SF2. The two unshared electron pairs take up more space than the shared pairs and so the shared pairs move closer together approximately degrees apart. Sf2 Molecular Geometry, Lewis Structure, Polarity and Bond AnglesĪtoms and Atomic Structure.

The material on this site can not be reproduced, distributed, transmitted, cached or otherwise used, except with prior written permission of Multiply.
